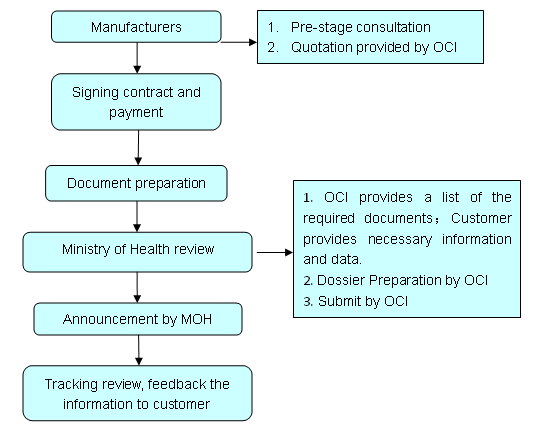
OCI service
a) Provision of pre-estimate of timing and costs for whole project;
b) Judgment analysis of Existing data and Provision of notification proposal and required tentative test proposal;
c) Dossier Preparation and Submit;
d) Tracking the whole review process of MOH review; and Providing of Proposal for Supplementary Material in time;
e) Feedback to customer with the latest progress of the registration and related results;
f) Provision of related translation according to the requirements of Ministry of Health;
g) Provision of an obligation of confidentiality upon the data provided.
The flow chart